=Reviewing concepts=
1. Which of the following is not an organic molecule?a. cellulose
b.surcrose
c.water
d. testosterone
answer= c. water
2. Which of the following terms includes all the other terms on this list?
a. polysaccharide
b. carbohydrate
c. monosaccharide
d. glycogen
answer= b. carbohydrate
3. Which term is most appropriate to describe a molecule that dissolves easily in water?
a. hydrocarbon
b. hydrophobic
c. hydrophilic
d. organic
answer= c. hydrophilic
4. Cholesterol is an example of what kind of molecule?
a. protein
b. lipid
c. amino acid
d. carbohydrate
answer= b. lipid
5. The 20 amino acids vary only in their
a. carboxyl groups
b. side groups
c. amino groups
d. lipid groups
answer= b. side groups
6. A specific reactant an enzyme acts upon is called the
a. catalyst.
b. sucrase
c. active site
d. substrate.
answer= d. substrate
7. An enzyme does which of the following?
a. adds heat to a reaction, speeding it up
b. lowers the activation energy of a reaction
c. cools a reaction, slowing it down
d. raises the activation energy of a reaction
answer= b.
8. Besides satisfying your hunger, why else might you consume a big bowl of pasta the night before a race?
- You need energy before the big race so pasta provides you carbohydrates that will help you run well in the race giving enough energy.
9. How are glucose, sucrose, and starch related?
-They are all made up of monosaccharides.
10.What are steroids? Describe two functions they have in cells.
- Steroid is a lipd molecule in which the carbon skeleton forms four fused rings and the function in circulating in our body as chemical signals and cause the male and female appearances.
11. How are polypeptides related to proteins?
- Polypeptides create all different kinds of proteins.
12. How does denaturation affect the ability of a protein to function?
- Denaturation affect proteins to change their shape and proteins do not function well.
*Applying Concepts
14. Analyzing Diagrams The reaction below shows two amino acids joining together.
a. One product of this reaction is represented by a question mark. WHich molecule is it?
- water molecule
b. WHat is this kind of reaction called? Explain
- Dehydration reaction takes place when a water molecule goes off and form a new bond with other polymers
c. If an amino acid were aded to this chain, at what two places could it attach?
- Amino acid can attach to either to the amino group of carboxyl group
15. Analyzing Graphs Use the graph to answer the questions below
a. At which temperature does enzyme A perform best? Enzyme B?
- Enzyme A: 38 degrees Enzyme B: 78 degrees
b. Knowing that one of these enzymes is found in humans and the other in thermophilic(heat-loving) bacteria, hypothesize which enzyme came from which organism.
- Enzyme A: human Enzyme B: thermophilic bacteria
c. Propose a hypothesis that explains why the rate of the reaction catalyzed by enzyme A slows down at temperatures above 40 degrees.
- Even though the temperature is above 40 degrees, the rate of the reaction slows down. That means the product will be shortly released and the reaction is about the end.

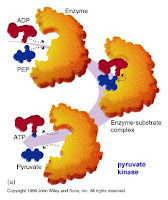 -Enzyme fits the shape of only particular reactant molecules
-Enzyme fits the shape of only particular reactant molecules









