=8.2 The light reactions convert light energy to chemical energy=
*Vocab
-wavelength=>distance between adjacent waves
-electromagnetic spectrum=> range of types of electromagnetic energy from gamma waves to radio waves
-pigment=>chemical compound that determines a substance's color
-paper chromatography=> laboratory technique used to observe the different pigments in a material
-photosystem=>cluster of chlorophyll and other molecules in a thylakoid
*Light Energy and Pigments
-Sunlight: form of electromagnetic energy
gamma rays-X-rays-UV-visible light-infrared-microwaves-radiowaves
>Pigments and Color
pigment choose the color of a substance
-three things happens to different wavelengths when it shines an object->absorbed, tranmitted, or reflected
-If a color is green, it is green because green is reflected from the object and other color is absorbed
>Identifying Chloroplast pigments
-We can observe different pigments in a green leaf by using laoratory tech called paper chromatography
-paper chromatography can separate and analyze the pigments in a leaf
*Harvesting Light energy
-We can watch the process occuring in a chloroplast as sunlight strikes a leaf.
-photosystems contains pigment molecules-the solar collectors
-Pigment's electrons gains energy as pigment molecules absorbs light energy
-chlorophyll a molecule get trapped by primary electron acceptor to be used to make ATP and NADPH
*Chemical Products of the Light Reactions
-light strikes photosystem and transfer excited electrons to the primary electron acceptor.
-electrons split water and releases oxygen
-excited electron goes through electron transport chain and pumps H+ions across the membrane to thylakoid.
-light excited electrons get ransferred to NADP+
*Concept Check
1.Explain why a leaf appears green
-leaf appears green because it reflects green light and absorb the other light colors
2.Describe what happens when a molecule of chlorophyll a absorbs light.
-when a molecule of chlorophyll a absorbs light it gets trapped by primary electron acceptor to produce ATP and NADPH.
3.Besides oxygen, what two molecules are produced by the light reactions?
-ATP & NADPH
4.Where in the chloroplast do the light reactions take place?
-It takes place in the thylakoid membrane

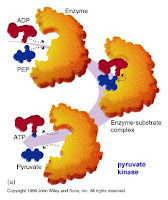 -Enzyme fits the shape of only particular reactant molecules
-Enzyme fits the shape of only particular reactant molecules




